Biotech Innovations Reshaping Diagnostics and Patient Care
Cutting-edge biotech breakthroughs are accelerating diagnostic speed and expanding treatment options across the US. From AI-powered screening to precision therapeutics, the field is delivering measurable gains in patient outcomes.
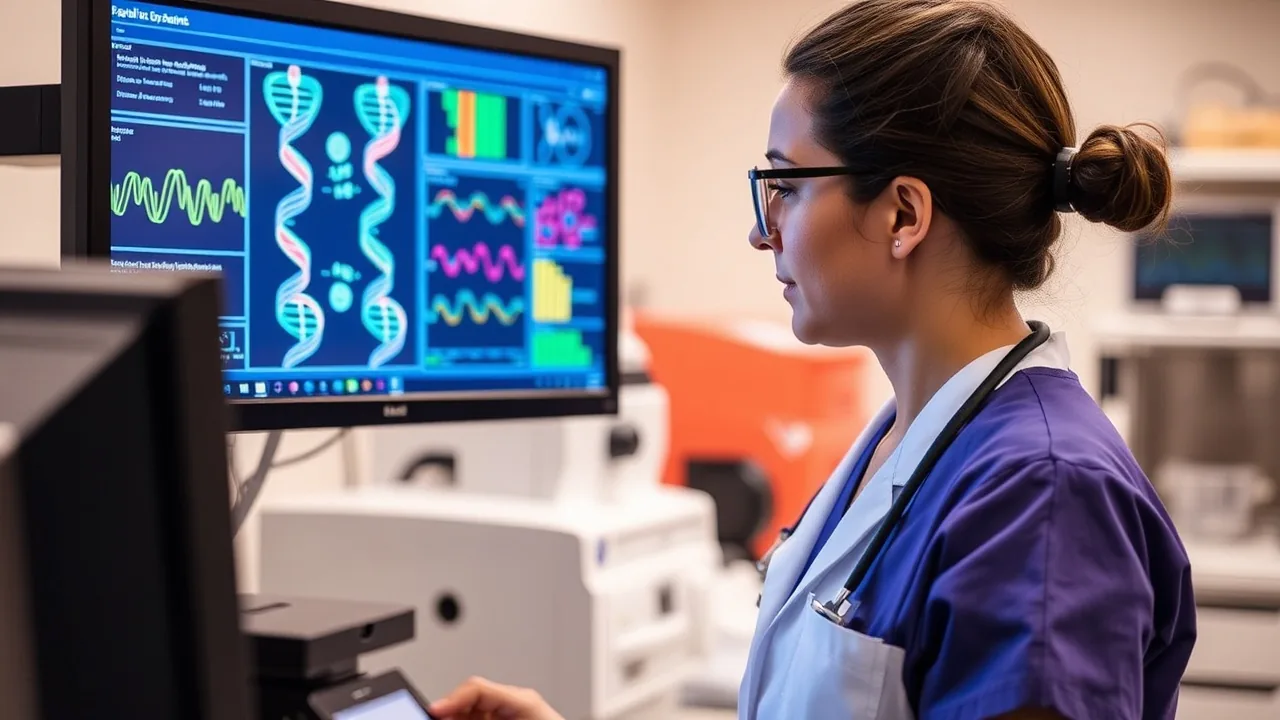
Scottish biotech firm Guthrie Technologies announced this week a new genetic screening platform that reduces diagnostic turnaround time from weeks to 48 hours, marking a significant leap in real-world clinical deployment. The system uses advanced sequencing paired with machine-learning interpretation, allowing hospitals across the Northeast and Midwest to identify rare genetic conditions faster and initiate treatment protocols ahead of previous timelines.
This announcement reflects a broader wave of innovation reshaping biotech across North America. The sector is no longer confined to research labs; it is now delivering tangible tools that clinicians use daily to improve outcomes for millions of patients.
"We're at an inflection point," says Dr. Margaret Chen, chief medical officer at the American Clinical Laboratory Association. "Five years ago, genetic testing was a last resort for complex cases. Today, it's becoming routine because turnaround times have dropped and costs have fallen by 60 to 70 percent."
Speed and Precision in Diagnostics
The race to accelerate diagnostic workflows has become central to modern medical technology. Laboratories and hospital networks are investing heavily in platforms that combine next-generation sequencing, bioinformatics, and cloud-based data processing. Guthrie's announcement underscores how automation and artificial intelligence are collapsing the timeline between sample collection and actionable results.
Key drivers of this shift include:
- Portable sequencing devices that work outside traditional lab settings
- Real-time data analysis using trained AI models
- Integration with electronic health records for seamless reporting
- Regulatory fast-track pathways for breakthrough technologies
Major hospital systems in Boston, Chicago, and San Francisco have already adopted similar platforms. Mayo Clinic reported in March 2024 that its deployment of AI-assisted diagnostic imaging cut radiologist review time by 35 percent while improving accuracy rates. Cleveland Clinic's genetics department now processes 40 percent more samples per month using automated sequencing pipelines than it did in 2022.
Cost remains a critical enabler. Early versions of these diagnostic systems required capital investments of $2 to $4 million per installation. Current-generation instruments start at $800,000, with per-test costs dropping from $3,000 to under $500 in high-volume centers.
Therapeutic Development Accelerating
Beyond diagnosis, genetic engineering and precision medicine are opening new therapeutic avenues at an unprecedented pace. CRISPR-based treatments for sickle cell disease and beta-thalassemia gained FDA approval in late 2023, moving from bench discovery to patient treatment in under a decade. CAR-T cell therapies, which reprogram immune cells to attack cancer, are now in use against seven distinct tumor types across US oncology centers.
Venture funding for clinical-stage biotech companies reached $18.7 billion in 2023, according to the Biotechnology Innovation Organization. While down from 2021 peaks, the figure reflects sustained confidence in therapeutic pipelines coming online over the next five years.
Regenerative medicine represents another frontier. Companies working on stem-cell derived tissues and organ-on-chip models are moving closer to clinical trials. University of Michigan researchers published results in February 2024 showing that lab-grown cartilage implants restored mobility in osteoarthritis patients over a 24-week period, with effects sustained at one-year follow-up.
Why Patient Care Is Changing Now
The convergence of three factors is driving rapid adoption of biotech innovation into routine patient care. First, regulatory agencies including the FDA have created expedited review pathways for technologies demonstrating clear clinical benefit. Breakthrough Device designation now covers over 150 active submissions, down from an average review time of 18 months to 6 months for qualifying applications.
Second, reimbursement models are shifting. Medicare and major insurers are moving from cost-per-procedure to value-based payment structures that reward diagnostic accuracy and treatment efficacy. This financial incentive encourages hospitals to adopt newer healthtech solutions faster.
Third, workforce availability is improving. Training programs in genetic counseling, bioinformatics, and laboratory genomics have expanded at 120 US universities since 2018, creating a talent pipeline that supports scale-up of new diagnostic and therapeutic platforms.
Patient demand is also rising. Online health communities and disease advocacy groups now routinely reference precision medicine options. Patients are asking clinicians about genetic testing and targeted therapies far more frequently than they did three years ago, pushing health systems to expand capacity.
The Guthrie announcement exemplifies this ecosystem in motion: a private firm deploying innovative medicine tools through hospital partnerships, regulatory approval enabling rapid scale, workforce training supporting implementation, and patient awareness driving uptake. These elements together are reshaping the speed and quality of care delivery across the country.
